
RFID
PEACE OF MIND
FOR YOUR PATIENTS



Give your patients the power of knowledge.
Women are increasingly understanding the importance of verifying their implant information. Asking questions such as:




These are real concerns, from real women.
And that’s why we created:
RFID, for peace of mind
Motiva® breast implants incorporate an optional radio frequency identification device (RFID), a small component embedded within the cohesive gel that enables non-invasive access to implant-specific information after implantation1-3 and verification of implanted devices, allowing you to support patients verifying their implant information.
This technology has been available around globally since 2014, and more than 2.5 million Motiva® Implants with Motiva’s first-generation RFID technology have been placed in the market, providing safety and reassurance to women of the implants they chose. There have been no reports of these devices leading to a clinical misdiagnosis or an inadequate assessment of implant integrity.4,5
Depending on the country, Motiva® Implants are optionally available with the RFID Qid® or Zen®, enabling you and your patients with real time non-invasive access to key device information and device verification.

Why did we invent RFID in implants?

Motiva® Implants with RFID*
Launched in 2014, RFID is an optional component that is embedded within the gel of Motiva® breast implants. It allows both patients and surgeons, after surgery, to easily access information about Motiva® implants.
Implants with microtransponders, like Qid® or Zen®, allow patients and health-care practitioners to check for counterfeits or devices different to the ones agreed upon for implantation, through immediate non-invasive verification of the exact device information including brand, model, size, serial number, and manufacturing date.1,2
*Qid® and Zen® are optional components and are not included in the entire Motiva® portfolio.



Want to know more?
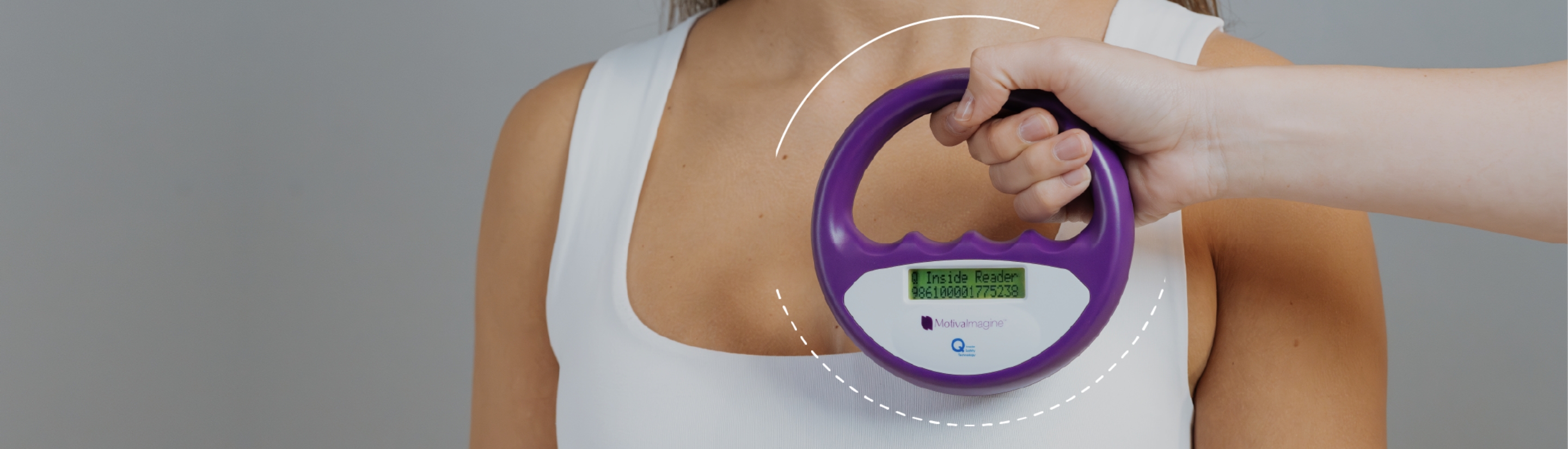
RFID in Motiva® implants is available in two formats: Qid® and Zen®. Both utilize passive RFID technology, meaning they do not contain a battery. Instead, their functionality relies on the connection to a proprietary handheld radiofrequency reader. When the reader is brought near the implant, it emits power that activates the component’s integrated circuit. This circuit then transmits a unique electronic serial number (ESN) via radio waves.1-3
Each ESN is unique to the individual implant and can be securely entered by a certified surgeon into the motiva.health professional database to retrieve specific implant and procedure information as needed.
Women are increasingly aware of the importance of knowing and verifying accurate information about their breast implants. Particularly in the unfortunate event of a device recall, manufacturer safety alerts or more recently, the emergence of breast implant-related diseases, such as breast implant-associated anaplastic large-cell lymphoma (BIA-ALCL)6.
Implant recalls from other companies have led some women to choose prophylactic explantation (preventative implant removal), even in the absence of adverse symptoms7 or device-related complications, driven through anxiety alone.8 The ability to easily access implant information has proven difficult for many women due to a lack of reliable breast implant registries, loss of medical records, loss of patient device cards, surgeon relocation/retiring and a significant increase in cosmetic tourism.9,10 Further, many women state they never received their implant information post-surgery.11
Qid® and Zen® enable women and healthcare professionals to access implant information non-invasively and verify breast implant details quickly from any* geographical location, overcoming previous challenges with implant traceability and potentially avoiding unnecessary surgical procedures. Moreover, verifying the previously implanted device prior to revision surgery can enhance the surgeon’s preoperative planning by ensuring knowledge of key implant characteristics, including surface type, volume, and shape. This information may also assist in planning capsule management by identifying the implant surface. The Food and Drug Administration (FDA) has further acknowledged the importance of radiologists being aware of the breast implant fill material(s), number of lumens, manufacturer, and model to support accurate interpretation and diagnosis during imaging surveillance.12,13 With RFID technology, this information can be easily accessed and verified.14
The safety of Qid® and Zen® has been tested in accordance with International Organization for Standardization15 (ISO), FDA testing guidelines,16 and by North American Science Associates (NAMSA) to ensure it meets all safety standards.17 These safety standards include the biological evaluation, chemical characterization, and toxicological assessment of Motiva® Implants with the optional microtransponder.17 These tests have confirmed that the technologies are safe for clinical use.18 For imaging considerations, see MRI section below.
The optional RFID technology featured in Motiva® implants is passive and responds only when prompted by the specific radiofrequency signal emitted by the handheld Motiva® Reader.19 It does not enable location tracking or allow remote monitoring, in contrast to “tracking” devices that continuously emit signals.20
The only information transmitted from Qid® and Zen® to the Motiva® Reader is the breast implants’ unique (ESN), which is accessible only by certified healthcare professionals when the Motiva® Reader is placed near the implant. Entering the ESN into the motiva.health database, allows access to specific implant information, including surface type, projection, base, volume, and manufacturing date. It does NOT provide access to any patient identifying information.
Women with Motiva® Implants featuring RFID technology (Qid® and Zen®) can safely undergo the following breast imaging modalities:21-23
- Mammography
- Computerized tomography (CT)
- X-ray,
- Ultrasound
- MRI, under specific conditions*
*Please refer to the implant’s Directions for Use, and refer to the question below.
Patients with Motiva® implants featuring RFID technology (Qid® or Zen®) are classified as MR Conditional and may safely undergo MRI under the conditions listed below. Motiva® implants without RFID technology are classified as MR Safe.2,24-27
- Magnetic Field Strength: MRI scanners must operate at 1.5-Tesla and 3-Tesla.*
- Maximum Spatial Gradient Magnetic Field: Up to 4,000-gauss/cm (40-T/m), extrapolated.
-
Specific Absorption Rate (SAR): Whole-body averaged SAR of 2 W/kg for:
- 15 minutes of scanning (i.e., per pulse sequence), or
- 60 minutes of continuous scanning (i.e., per pulse sequence or back-to-back sequences/series without breaks) in the Normal Operating Mode.
Patients with Motiva® implants featuring RFID technology should notify their MRI technologist of the RFID presence and provide access to the information provided in the device IFU and/or information on the implant ID card.
*These are considered standard for clinical imaging in most radiology departments worldwide.28 While 7-Tesla scanners may be available in specialized settings, they are typically reserved for research purposes.2
Both Qid® and Zen® are embedded within the highly-cohesive silicone gel,30
on the posterior portion of the implant. Post-market surveillance data over the last decade has shown no
reported cases of RFID migration into the surrounding breast tissue.31
In the event of an implant rupture, a complete chemical characterization and biocompatibility assessment of
Qid® and Zen® have been conducted, followed by a subsequent toxicological
evaluation, and the results demonstrated that both technologies are non-cytotoxic and safe for clinical
use.32
How to verify patient’s Motiva Implants® with RFID?
Watch the video to learn how you can aid
patients to verify their implants.

How to verify if patient´s Motiva Implants have Qid® or Zen® ?
You can confirm if Motiva® implants include RFID technology such as Qid® or Zen® via the catalogue or reference number on the Motiva® Implant Card (as seen in the image).
- If the number ends with the letter “Q”, your implants include Qid®.
- If the number ends with the letter “Z”, your implants include Zen®.
- If there are no letters present, it means that RFID technology is not included in your breast implants.
References
- Establishment Labs. DDD-001: Device Description Document for Sterile Silicone Breast Implants Motiva® implants Round SmoothSilk®/SilkSurface® Plus. Data on file.
- Establishment Labs, DDD-002: Device Description Document for Sterile Silicone Breast Implants Motiva® implants Ergonomix® Round SmoothSilk®/SilkSurface®. Data on File
- Establishment Labs. DDD-006: Device Description Document for Motiva Implants Ergonomix2. Data on file.
- Establishment Labs, Compliance Quest Complaints Report October 2025. Data in File
-
Establishment Labs. PMS Abstract 2025: 15 Years of Motiva® Data – Post‑Market
Surveillance Report. Published October 2025. Accessed January 27, 2026.
https://www.establishmentlabs.com/wp-content/uploads/2025/10/PMS-Abstract-2025.pdf - U.S. Food and Drug Administration. Medical Device Reports of Breast Implant-Associated Anaplastic Large Cell Lymphoma. Published February 6, 2025. Accessed February 3, 2025 https://www.fda.gov/medical-devices/breast-implants/medical-device-reports-breast-implant-associated-anaplastic-large-cell-lymphoma
- PIP Breast Implants Situation Update. Agence nationale de sécurité de medicament et des produit de santé report. April 2013. Pg.15.http//:ansm.sante.fr/content/download/.../Update-PIP-UKjune2013.pd
- SCENIHR (Scientific Committee on Emerging and Newly Identified Health Risks), Scientific opinion on the Safety of Poly Implant Prothèse (PIP) Silicone Breast Implants (2013 update), 12 May 2014.
- Sang Eun Nam, Bum Sik Bang, Eun Kyoung Lee, et al. Use of High-Resolution Ultrasound in Characterizing a Breast Implant and Detecting a Rupture of the Device. Plastic and reconstructive surgery/PSEF CD journals. 2023;152(1):39-43. doi:https://doi.org/10.1097/prs.0000000000010201
- Fabio, Panagiotakos D, Firmani G, Michail Sorotos. BIA-ALCL Epidemiological Findings From a Retrospective Study of 248 Cases Extracted From Relevant Case Reports and Series: A Systematic Review. Aesthetic Surgery Journal. 2022;43(5):545-555.doi: https://doi.org/10.1093/asj/sjac3124
- Glicksman C. et al. The Aesthetic One App Revolutionizes Implant Registration and Creates the Connected Patient, Aesthetic Surgery Journal, Volume 42, Issue 7, July 2022, Pages NP447–NP450, https://doi.org/10.1093/asj/sjac073
- Nelson MT, Meisamy S. High Risk Breast Cancer Patient with Silicone Breast Implant and Q Inside SafetyTM Micro Transponder. Open journal of medical imaging. 2019;09(04):52-57. doi:https://doi.org/10.4236/ojmi.2019.94005
- Munhoz AM, Chala L, de Melo G, Marques AA, Tucunduva T, Gemperli R. Usefulness of radiofrequency identification device in diagnosing rotation of Motiva SmoothSilk implants after augmentation mammoplasty. Plast Reconstr Surg Glob Open 2019;7:e2497; doi:10.1097/GOX.0000000000002497
- Huong T Le-Petross, MD, FRCPC and others, Assessment, Complications, and Surveillance of Breast Implants: Making Sense of 2022 FDA Breast Implant Guidance, Journal of Breast Imaging, Volume 5, Issue 3, May/June 2023, Pages 360–372, https://doi.org/10.1093/jbi/wbad029
- International Organization for Standardization. ISO 10993-1:2018: Biological evaluation of medical devices — Part 1: Evaluation and testing within a risk management process. Published August 2018. Accessed February 4, 2026. https://www.iso.org/standard/68936.html
- U.S. Food and Drug Administration. Use of International Standard ISO 10993-1, "Biological evaluation of medical devices – Part 1: Evaluation and testing within a risk management process." Guidance for Industry and Food and Drug Administration Staff. Published September 8, 2023. Accessed February 4, 2026.https://www.fda.gov/regulatoryinformation/search-fda-guidance-documents/use-international-standard-iso-10993-1-biological-evaluation-medical-devices-part-1-evaluation-and
- International Organization for Standardization. ISO/TS 21726:2019: Biological evaluation of medical devices — Application of the threshold of toxicological concern (TTC) for assessing biocompatibility of medical device constituents. Published February 2019. Accessed February 4, 2026. https://www.iso.org/standard/71514.html
- Establishment Labs. BER-001002: Biological Evaluation Report (BER) for Microtransponder. Data on file.
- Nelson MT, Brattain KA, Williams JM. Does Electronic Identification Enablement for Silicone Gel Implants Impact Patient Safety? J Surg OpenAccess. 2018;4(1). doi: 10.16966/2470-0991.162
- Profetto L, Gherardelli M, Iadanza E. Radio Frequency Identification (RFID) in health care: where are we? A scoping review. Health and Technology. 2022;12(5):879-891. doi:https://doi.org/10.1007/s12553-022-00696-1
- Establishment Labs. TS-001146: Safety and Performance of the Motiva Implants® with microtransponder(DRW-001053) exposed to Magnetic Resonance Environment. Data on file.
- Meisamy S, Nelson MT. The Effects of Q Inside Safety Technology Micro Transponder on Routine Breast Implant Imaging. Open Journal of Medical Imaging. 2019;09(02):19-31. doi:https://doi.org/10.4236/ojmi.2019.92002
- Establishment Labs. TS-001298: Technical Study Report Artifact assessment when comparing Ferrite Microtransponder (Qid®) vs Non-Ferrite Microtransponder (Zen®) when submitting the samples to various medical imaging modalities. Data on file.
- Establishment Labs. VAL-16-018.R: Report for the Safety and Performance of the Motiva Implants® with Qid® exposed to an MRI Environment. Data on file.
- Establishment Labs. TS-001146: Safety and Performance of the Motiva® implants with microtransponder (DRW-001053) exposed to Magnetic Resonance Environment. Data on file.
- Shrivastava D, Utecht L, Tian J, Hughes J, and Vaughan JT, “In Vivo Radiofrequency Heating in Swine in a 3T (123.2 MHz) Birdcage Whole-Body Coil,” Magn Reson Med., 2014, Vol. 72, No. 4, pp. 1141–50.
- Tian J, Shrivastava D, “In Vivo Radiofrequency Heating in a 3T MRI Scanner,” 2018, in Theory and Applications of Heat Transfer in Humans, Editor Shrivastava D, John Wiley and Sons, Chichester, West Sussex, UK.
- Medicines and Healthcare products Regulatory Agency. Guidance on MRI Safety. UK Government; 2021. Accessed February 3, 2026. https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/958486/MRI_guidance_2021-4-03c.pdf
- Okada T, Akasaka T, Thuy DH, Isa T. Safety for Human MR Scanners at 7T. Magnetic Resonance in Medical Sciences. 2022;21(4):531-537. doi:https://doi.org/10.2463/mrms.rev.2021-0063
- Aitzetmuller-Kleitz ML, Yang S, Wiebinghaus P, Wellenbrock S, Ozturk M, Kuckelhaus M et al. Complication rates after breast surgery with the Motiva Smooth SilkSurface silicone gel implants- A systematic review and meta-analysis. Clin. Med. 2023, 12,1881. doi: 10.3390/jcm12051881
- Establishment Labs®, Post-Market Surveillance Results 15-Year Report.
- Establishment Labs. BER-001002: Biological Evaluation Report (BER) for Microtransponder. Data on file.
Disclaimer:
The information provided is solely for informational purposes. The manufacturer affirms that the use of this technology complies with current regulatory and safety standards. All the details related to the RFID, including specified conditions of use for MRI environment, can be found in the Directions for Use document attached to your Motiva product and our Information for the Patient document. The information presented herein does not constitute medical advice and should not replace consultation with qualified healthcare professionals.

